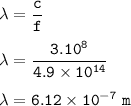The wavelength-λ = 6.12 x 10⁻⁷ m
Further explanation
Radiation energy is absorbed by photons
The energy in one photon can be formulated as
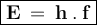
Where
h = Planck's constant (6,626.10⁻³⁴ Js)
f = Frequency of electromagnetic waves
f = c / λ
c = speed of light
= 3.10⁸ m/s
λ = wavelength
The wavelength of radiation with a frequency of 4.9 ×10¹⁴/s :