Answer: 226 Subscript 88 Baseline Upper R a right arrow Superscript 222 Subscript 86 Baseline Upper R n + Superscript 4 Subscript 2 Baseline Upper H e
Step-by-step explanation:
Alpha decay : When a larger nuclei decays into smaller nuclei by releasing alpha particle. In this process, the mass number and atomic number is reduced by 4 and 2 units respectively.
The general representation of alpha decay reaction is:
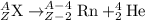
Representation of Radium decays to form Radon
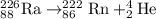
Thus 226 Subscript 88 Baseline Upper R a right arrow Superscript 222 Subscript 86 Baseline Upper R n + Superscript 4 Subscript 2 Baseline Upper H e represents alpha decay.