Answer:
The answer is
1479.60 mL
Step-by-step explanation:
In order to calculate the volume needed we use the formula
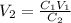
where
C1 is the concentration of the stock solution
V1 is the volume of the stock solution
C2 is the concentration of the diluted solution
V2 is the volume of thevdiluted solution
From the question
C1 = 1.45 M
V1 = 25 mL
C2 = 0.0245 M
So we have
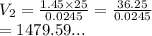
We have the final answer as
1479.60 mL
Hope this helps you