Answer:
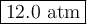
Step-by-step explanation:
The volume and amount of gas are constant, so we can use Gay-Lussac’s Law:
At constant volume, the pressure exerted by a gas is directly proportional to its temperature.
\dfrac{p_{1}}{T_{1}} = \dfrac{p_{2}}{T_{2}}
Data:
p₁ = 9.00 atm; T₁ = 28.0 °C
p₂ = ?; T₂ = 129.0 °C
Calculations:
1. Convert the temperatures to kelvins
T₁ = (28.0 + 273.15) K = 301.15
T₂ = (129.0 + 273.15) K = 402.15
2. Calculate the new pressure
