Option B, C and D are correct.
Step-by-step explanation:
Carbon is a non metallic element with the atomic number 6 and mass number 12. Whereas oxygen is also a non metallic element with the atomic number 8 and mass number of 16.
In carbon monoxide, the mass ratio of oxygen to carbon is 16:12 =1.33. This ratio is same for every sample of carbon monoxide, because carbon monoxide has the universal formula of CO.
Similarly In carbon dioxide , the mass ratio of oxygen to carbon is 32:12 =2.667. This ratio is same for every sample of carbon dioxide, because carbon dioxide has the universal formula of
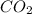 .
.
Even we can see that the mass ratio of oxygen to carbon in carbon dioxide is just twice the mass ratio of oxygen to carbon in carbon monoxide, because of presence of twice as much as oxygen per molecule of carbon dioxide than carbon monoxide.