Answer:

Step-by-step explanation:
Since we are given the mass, specific heat, and temperature, we should use the following formula for heat energy.
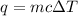
The mass of the aluminum is 26.3 grams. Its specific heat is 0.930 Joules per gram degree Celsius. We need to find the change in temperature.
- The change in temperature is the difference between the initial temperature to the final temperature.
- The temperature changes from 23.0°C to 67.0°C, so the initial is 23 degrees and the final is 67 degrees.
- ΔT= final temperature - initial temperature
- ΔT= 67°C - 23°C
- ΔT= 44°C
Now we know all the values.
- m= 26.3 g
- c= 0.930 J/g °C
- ΔT= 44°C
Substitute the values into the formula.
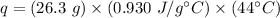
Multiply the first two numbers together. The units of grams cancel.
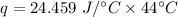
Multiply again. This time, the units of degrees Celsius cancel.
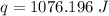
1076.196 Joules of heat will be absorbed by the piece of aluminum.