Answer:
D. Forces between molecules
Step-by-step explanation:
Specific heat capacity of water can be defined as the amount of heat a gram of water must lose or absorb in order to change its temperature by a degree Celsius. It is measured in Joules per kilogram per degree Celsius (J/kg°C). Generally, the specific heat capacity of water is 4.182J/kg°C and is the highest among liquids.
Mathematically, the specific heat capacity of a substance is given by the formula;
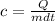
Where;
Q represents the heat capacity or quantity of heat.
m represents the mass of an object.
c represents the specific heat capacity of water.
dt represents the change in temperature.
Cohesion is a property of water and it typically refers to the attraction between molecules of water which holds them together.
In Science, the property which helps to explain differences in the specific heat capacities of two substances is the forces between molecules.
This ultimately implies that, the more closely bonded the atoms of a substance are, the higher or greater would be the substance's specific heat capacity. Thus, it varies for the various states of matter i.e solid, liquid and gas.