An element can have multiple isotopes. Isotopes correspond to variations of the same element with respect to the number of neutrons in its nucleus. the number they give us, 35 and 37 correspond to the mass number of chlorine. The percentage will be how abundant the element is.
To find the average atomic mass we must multiply the mass of the isotope by its respective percentage of abundance and add these two results.
So, the average atomic mass of Cl will be:
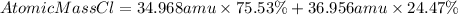
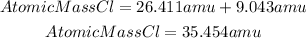
Answer: the average atomic mass of chlorine is 35.454 amu