INFORMATION:
We know that:
- One brand of laundry bleach is an aqueous solution containing 4.55% sodium hypochlorite (NaOCI) by mass
- The density is 1.02 g/mL
And we must find the molarity of this solution
STEP BY STEP EXPLANATION:
To find the molarity, we need to use that
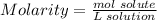
So, first we must calculate mol of the solute
To find mol of the solute we need to use that the brand is an aqueous solution containing 4.55% sodium hypochlorite (NaOCI).
In this case, sodium hypochlorite is the solute, so we can represent the percentage as
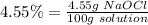
mol of solute:
Now, using the molar mass of sodium hypochlorite we can convert grams to mol
molar mass of sodium hypochlorite: 74.4 g/mol
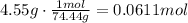
So, mol solute = 0.061 mol
L of solution:
Then, using the density we can convert g to mL and then mL to L
Density: 1.02 g/mL
1 L = 1000mL
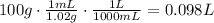
so, L solution = 0.098 L
Finally, replacing in the initial formula
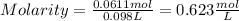
ANSWER:
C) 0.623