Answer:
Moles of hydrogen formed = 3.5 moles
Step-by-step explanation:
Given that:-
Moles of aluminium= 4.0 mol
Moles of hydrogen bromide = 7.0 mol
According to the reaction:-
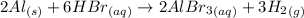
2 moles of aluminum react with 6 moles of hydrogen bromide
1 mole of aluminum react with 6/2 moles of hydrogen bromide
4 moles of aluminum react with (6/2)*4 moles of hydrogen bromide
Moles of hydrogen bromide = 12 moles
Available moles of hydrogen bromide = 7.0 moles
Limiting reagent is the one which is present in small amount. Thus, hydrogen bromide is limiting reagent. (7.0 < 12)
The formation of the product is governed by the limiting reagent. So,
6 moles of hydrogen bromide on reaction forms 3 moles of hydrogen
1 moles of hydrogen bromide on reaction forms 3/6 moles of hydrogen
7 moles of hydrogen bromide on reaction forms (3/6)*7 moles of hydrogen
Moles of hydrogen formed = 3.5 moles