Step-by-step explanation:
The given data is as follows.
Concentration of standard NaOH solution = 0.1922 M
Let the original acid solution concentration be x.
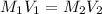
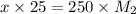
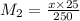
= 0.1 x M
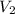 = 10.00 mL (given)
= 10.00 mL (given)
The reaction equation is as follows.
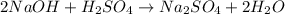
Concentration × Volume of
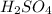 = Concentration × Volume of NaOH
= Concentration × Volume of NaOH
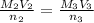
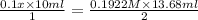
x = 1.314 M
Therefore, we can conclude that the concentration of the original sulfuric acid solution is 1.314 M.