Answer: The number of atoms of bromine present in given number of mass is

Step-by-step explanation:
To calculate the number of moles, we use the equation:
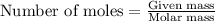
Given mass of bromine = 1.37 g
Molar mass of bromine = 79.904 g/mol
Putting values in above equation, we get:
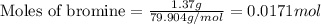
According to mole concept:
1 mole of an element contains
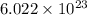 number of atoms.
number of atoms.
So, 0.0171 moles of bromine will contain =
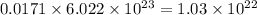 number of bromine atoms.
number of bromine atoms.
Hence, the number of atoms of bromine present in given number of mass is
