Answer: The given isotope of tin has 50 protons and 69 neutrons.
Step-by-step explanation:
Atomic number is defined as the number of protons or number of electrons that are present in neutral atom. It is represented as Z.
Atomic number = Number of protons = Number of electrons
Mass number is defined as the sum of number of protons and number of neutrons. It is represented as A.
Mass number = Number of protons + Number of neutrons
We are given:
An isotope having representation
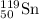
Mass number of Sn = 19
Atomic number = 50
Number of neutrons = Mass number - Atomic number = 119 - 50 = 69
Hence, the given isotope of tin has 50 protons and 69 neutrons.