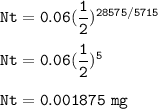The sample of Carbon-14 after 28,575 years=0.001875 mg
Further explanation
General formulas used in decay:
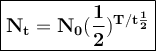
T = duration of decay
t 1/2 = half-life
N₀ = the number of initial radioactive atoms
Nt = the number of radioactive atoms left after decaying during T time
Carbon-14 has a half-life of 5715 years, so t1/2=5715 years
A sample today contains 0.060 mg of carbon-14, so No=0.06 mg, then :