The molarity of an acetic acid solution : 0.321 M
Further explanation
Titration is a procedure for determining the concentration of a solution by reacting with another solution that is known to be concentrated (usually a standard solution). Determination of the endpoint/equivalence point of the reaction can use indicators according to the appropriate pH range
Reaction
HC₂H₃O₂(aq) + NaOH(aq) → H₂O(l) + NaC₂H₃O₂(aq)
Molarity NaOh=0.212
volume NaOH=25 ml=0.025 L
mol NaOH =
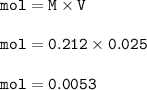
mol ratio NaOH : HC₂H₃O₂ from the equation = 1 : 1, so mol HC₂H₃O₂ :
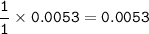
The molarity of acetic acid HC₂H₃O₂ :

Or you can use titration formula :
