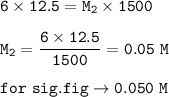The molarity of the dilution solution is 0.050 M
Further explanation
Molarity is a way to express the concentration of the solution
Molarity shows the number of moles of solute in every 1 liter of solute or mmol in each ml of solution
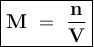
Where
M = Molarity
n = Number of moles of solute
V = Volume of solution
Dilution formula :
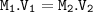
M₁=6 M
V₁=12.5 ml
V₂=1.5 L=1500 ml