Answer:
Oxide of manganese, Potassium permanganate, Manganese bromide and manganese oxide.
Step-by-step explanation:
1. Manganese dioxide
Its chemical formula is
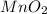 . It is +4 oxidation state. It is a black solid which is fairly stable.
. It is +4 oxidation state. It is a black solid which is fairly stable.
2. Potassium permanganate
Its chemical formula is
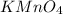 . It is commonly used in the laboratories as oxidizing agent. They appear black in color.
. It is commonly used in the laboratories as oxidizing agent. They appear black in color.
3 Manganese bromide
Its chemical formula is
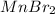
4. Oxides of manganese
Chemical formula is
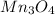
The most stable state of Manganese is +2 oxidation state.