Answer:
I = 1.05x10⁻³ A
Step-by-step explanation:
By definition, an electric current is the rate of charge flow at a given time:
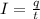
Where:
q: is the electrons charge = 1.602x10⁻¹⁹ C
t: is the time
In a circular motion, the time is given by:
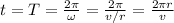
Where:
ω: is the angular speed = v/r
v: is the speed = 2.19x10⁶ m/s
r: is the radius = 5.29x10⁻¹¹ m
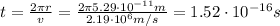
Now, the effective current is:
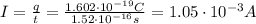
Therefore, the effective current associated with this orbiting electron is 1.05x10⁻³ A.
I hope it helps you!