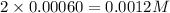Answer: The equilibrium concentration of
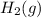 at 700 degrees Celsius is 0.0012 M
at 700 degrees Celsius is 0.0012 M
Step-by-step explanation:
Equilibrium constant is defined as the ratio of concentration of products to the concentration of reactants each raised to the power their stoichiometric ratios. It is expressed as
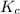
Moles of
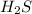 = 0.29 mole
= 0.29 mole
Volume of solution = 3.0 L
Initial concentration of
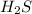 =
=
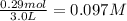
The given balanced equilibrium reaction is,
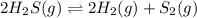
Initial conc. 0.097 M 0M 0M
At eqm. conc. (0.097-2x) M (2x) M (x) M
The expression for
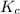 is written as:
is written as:
![K_c=([H_2]^2* [S_2])/([H_2S]^2)](https://img.qammunity.org/2021/formulas/chemistry/college/fic1fyq0dlvonn8p1j1f6xlebdiliihjfa.png)
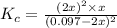
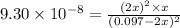
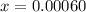
Equilibrium concentration of
![[H_2]](https://img.qammunity.org/2021/formulas/chemistry/college/g5lz38oh5k4n4u2tsea0idxcfrr4i5e121.png) = 2x=
= 2x=