Answer:
The answer is 55.54 K
Step-by-step explanation:
In order to find out the final temperature we use the formula for Charles's law which is
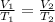
where
V1 is the initial volume
T1 is the initial temperature in Kelvin
V2 is the final volume
T2 is the final temperature in Kelvin
Since we are finding the final temperature
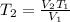
rom the question
V1 = 5.5 L
T1 = -38°C = -38 + 273 = 235 K
V2 = 1.30 L
We have
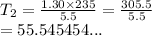
We have the final answer as
55.54 K
Hope this helps you