Answer:
Step-by-step explanation:
From the given information:
The density of O₂ gas =
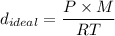
here:
P = pressure of the O₂ gas = 310 bar
=
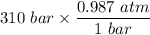
= 305.97 atm
The temperature T = 415 K
The rate R = 0.0821 L.atm/mol.K
molar mass of O₂ gas = 32 g/mol
∴
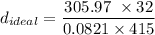
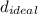 = 287.37 g/L
= 287.37 g/L
To find the density using the Van der Waal equation
Recall that:
the Van der Waal constant for O₂ is:
a = 1.382 bar. L²/mol² &
b = 0.0319 L/mol
The initial step is to determine the volume = Vm
The Van der Waal equation can be represented as:
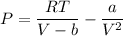
where;
R = gas constant (in bar) = 8.314 × 10⁻² L.bar/ K.mol
Replacing our values into the above equation, we have:
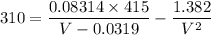
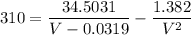
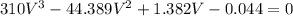
After solving;
V = 0.1152 L
∴
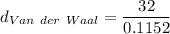
 = 277.77 g/L
= 277.77 g/L
We say that the repulsive part of the interaction potential dominates because the results showcase that the density of the Van der Waals is lesser than the density of ideal gas.