Answer:
The answer is
12,574,750.83 moles
Step-by-step explanation:
In order to find the moles of CO2 we use the formula
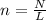
where n is the number of moles
N is the number of entities
L is the Avogadro's constant which is
6.02 × 10²³ entities
From the question
N = 7.57 × 10^30 molecules of CO2
We have
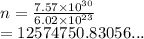
We have the final answer as
12,574,750.83 moles
Hope this helps you