Answer:
Step-by-step explanation:
PCl₅ ⇄ PCl₃ + Cl₂
1 mole 1 mole 1 mole
molecular weight of PCl₅ = 208.5
molecular weight of PCl₃ = 137.5
molecular weight of Cl₂ = 71
moles of PCl₅ = .0185
moles of PCl₃ = .0924
moles of Cl₂ = .1873
Total moles = .2982 moles
mole fraction of PCl₅ = .062
mole fraction of PCl₃ = .31
mole fraction of Cl₂ = .628
If total pressure be P
partial pressure of PCl₅ = .062 P
partial pressure of PCl₃ = .31 P
partial pressure of Cl₂ = .628 P
Kp = .31 P x .628 P / .062 P
= 3.14 P
To calculate Total pressure P
PV = nRT
P x 4 x 10⁻³ = .2982 x 8.31 x 600
P = 371.7 x 10³
= 3.717 x 10⁵ Pa
Kp = 3.14 P = 3.14 x 3.717 x 10⁵ Pa
= 11.67 x 10⁵ Pa
Kp = Kc x
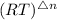
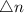 = 1
= 1
11.67 x 10⁵ = Kc x
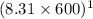
Kc = 234