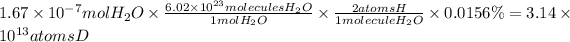Answer:
1.67 × 10⁻⁷ mol H₂O
3.14 × 10¹³ atoms deuterium
Step-by-step explanation:
How many moles of water were injected?
We have 3.00 μg of gaseous H₂O. To calculate the number of moles of water we will use the following relationships:
- 1 g = 10⁶ μg
- The molar mass of water is 18.02 g/mol.
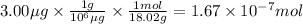
If the sample contains 0.0156% deuterium, how many deuterium atoms were injected?
We will use the following relationships:
1 mole of water contains 6.02 × 10²³ molecules of water.
There are 2 hydrogen atoms per water molecule.
0.0156% of hydrogen atoms are deuterium atoms.