Answer and Explanation:
The octet rue depicts the overall trend of chemical bonding in which atoms of an elements would seek to attain stability by filling out their valence shell with eight (8) electrons, giving it the same electronic configuration as a noble gas (specifically Argon).
Hence Carbon with 4 valence electrons (2, 4 electronic configuration) would take on four bromine atoms with 7 valence electrons (2, 8, 4 electronic configuration) each, giving each Br atom the single electron it needs to achieve octet stability.
Carbon tetrabromide
Chemical formula:
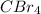
I hope this is a clear explanation