Answer:
≈ 0.70 g/cm³ (answer rounded up to 2 decimal places)
Step-by-step explanation:
The mass of empty cylinder is 20g
The mass of water filling the cylinder = 30g - 20g = 10g
The mass of unknown liquid filling the cylinder = 27g - 20g = 7g
Density of water = 997 kg/m³
Converting this density to g/cm³ we get;
1kg = 1000g , 1m³ = 1000000cm³
So density =
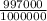 g/cm³ = 0.997 g/cm³
g/cm³ = 0.997 g/cm³
So the volume of water occupied by 10g is;
10 × 0.997 = 9.97 cm³
This volume is also occupied by 7g of the unknown liquid.
Density = mass/volume
Density of the unknown liquid = 7g ÷ 9.97cm³ = 0.702106319 g/cm³ ≈ 0.70 g/cm³ (answer rounded up to 2 decimal places)