Answer:
heptan-2-one
Step-by-step explanation:
In this case, the final product would be a ketone: heptan-2-one. To understand why this molecule is produced we have to check the reaction mechanism.
The first step is the protonation of the triple bond to produce the more stable carbocation (a secondary one) by the action of sulfuric acid
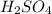 . The next step is the attack of water to the carbocation to produce a new bond between C and the O, producing a positive charge in the oxygen. Then, a deprotonation step takes place to produce an enol. Finally, we will have a rearrangement (keto-enol tautomerism) to produce the final ketone.
. The next step is the attack of water to the carbocation to produce a new bond between C and the O, producing a positive charge in the oxygen. Then, a deprotonation step takes place to produce an enol. Finally, we will have a rearrangement (keto-enol tautomerism) to produce the final ketone.
See figure 1
I hope it helps!