Answer:
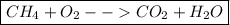
Step-by-step explanation:
Part 1: Naming compound formulas given the names
Step 1. Methane's formula is
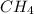 .
.
Step 2: Oxygen is a diatomic molecule (it exists bonded to itself for stability purposes), so by itself in chemical equations, it is written as
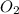 .
.
Step 3: Carbon dioxide is the molecular compound of one atom of carbon and two atoms of oxygen →
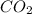 .
.
Step 4: Water is the common name of the compound of two hydrogen atoms and one oxygen atom →
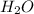 .
.
Part 2: Writing the skeleton equation
Step 1: Use the determined formulas for the reactants and plug them into the equation. We are told that methane burns in oxygen -- hinting at a combustion reaction. Therefore, we may infer that these are the reactants that yield the products.
Skeleton equations are written with the reactant(s) on the left -- if there are several, they are separated by an addition symbol (+).
With this information, we may begin our equation:
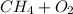 , where
, where
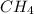 is methane and
is methane and
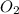 is the diatomic molecule of oxygen.
is the diatomic molecule of oxygen.
Step 2: Use the determined formulas for the products and plug them into the equation. We are told that the methane burns in oxygen to produce carbon dioxide and water. Hence, we can separate these two as we did with the reactants.
Now, our products side of the reaction will look like this:
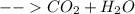 , where
, where
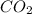 is carbon dioxide and
is carbon dioxide and
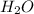 is water.
is water.
Step 3: Write the final equation. All you must do after determining both sides of the equation is simply push them together. Place the reactant side of the equation on the left and the product side of the equation on the right.
This gives us our final equation,
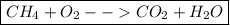 .
.
Because the problem asks for the unbalanced equation, we do not need to take any further steps of balancing the equation.