Answer:
See explanation
Step-by-step explanation:
For this question, we have to remember the effect of an atom with high electronegativity as "Br". If the "Br" atom is closer to the carboxylic acid group (COOH) we will have an inductive effect. Due to the electronegativity of Br, the electrons of the C-H bond would be to the Br, then this bond would be weaker and the compound will be more acid (because is easier to produce the hydronium ion
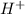 ).
).
With this in mind, for A in the last compound, we have 2 Br atoms near to the acid carboxylic group, so, we will have a high inductive effect, then the C-H would be weaker and we will have more acidity. Then we will have the compound with only 1 Br atom and finally, the last compound would be the one without Br atoms.
In B, the difference between the molecules is the position of the "Br" atom in the molecule. If the Br atom is closer to the acid group we will have a higher inductive effect and more acidity.
See figure 1
I hope it helps!