Answer:
0.49 mol
Step-by-step explanation:
Step 1: Write the balanced equation
Mg + 2 HCI ⇒ MgCl₂ + H₂
Step 2: Calculate the moles corresponding to 12 g of Mg
The molar mass of Mg is 24.31 g/mol.
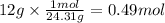
Step 3: Calculate the moles of H₂ produced by 0.49 moles of Mg
The molar ratio of Mg to H₂ is 1:1. The moles of H₂ produced are 1/1 × 0.49 mol = 0.49 mol.