Answer:
0.374 L
Step-by-step explanation:
Step 1: Given data
Mass of silver nitrate (solute): 15.89 g
Molarity of the solution: 0.250 M
Step 2: Calculate the moles of silver nitrate
The molar mass of silver nitrate is 169.87 g/mol.
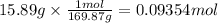
Step 3: Calculate the volume of the solution
The molarity is equal to the moles of solute divided by the liters of solution.
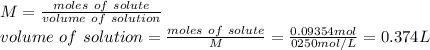
Due to the significant figures rules, we keep 3 significant figures.