Answer:
c = 0.38 J/g °C
Explanation:
We have,
Mass of metal X is 95.4 g
Initial temperature was 25°C and final temperature is 48°C.
The metal absorbs 849 J of heat
The heat absorbed due to change in temperature is given by :
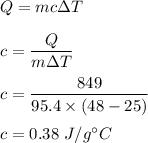
So, the specific heat of metal X is 0.38 J/g °C.