Answer: pH=2.38
Step-by-step explanation:
To calculate the pH, let's first write out the equation. Then, we will make an ICE chart. The I in ICE is initial quantity. In this case, it is the initial concentration. The C in ICE is change in each quantity. The E is equilibrium.
HCOOH ⇄ H⁺ + HCOO⁻
I 1.0M 0 0
C -x +x +x
E 1.0-x x x
For the steps below, refer to the ICE chart above.
1. Since we were given the initial of HCOOH, we can fill this into the chart.
2. Since we were not given the initial for H⁺ and HCOO⁻, we will put 0 in their place.
3. For the change, we need to add concentration to the products to make the reaction reach equilibrium. We would add on the products and subtract from the reactants to equalize the reaction. Since we don't know how much the change in, we can use variable x.
4. We were given the Kₐ of the solution. We know
![K_(a) =(product)/(reactant)=([H^+][HCOO^-])/([HCOOH])](https://img.qammunity.org/2021/formulas/chemistry/college/qxcbppomfg25z8cpc7jt9ltxw5ecuji1z3.png) .
.
5. The problem states that the Kₐ=1.8×10⁻⁴. All we have to so is to plug it in and to solve for x.
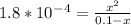
6. Once we plug this into the quadratic equation, we get x=0.00415.
7. The equilibrium concentration of [H⁺]=0.00415. pH is -log(H⁺).
-log(0.00415)=2.38
Our pH for the weak acid solution is 2.38.