Answer:
185.49 grams of Zinc would react with 454g (1lb) of copper sulfate
Step-by-step explanation:
Yo know the following balanced reaction:
CuSO₄(aq)+ Zn(s) →Cu(s) + ZnSO₄(aq)
You can see that by stoichiometry of the reaction (that is, the relationship between the amount of reagents and products in a chemical reaction), the following amounts of reagents and products are part of the reaction:
- CuSO₄: 1 mole
- Zn: 1 mole
- Cu: 1 mole
- ZnSO₄: 1 mole
Being:
- Cu: 63.54 g/mole
- S: 32 g/mole
- O: 16 g/mole
- Zn: 65.37 g/mole
the molar mass of the compounds participating in the reaction is:
- CuSO₄:63.54 g/mole + 32 g/mole + 4*16 g/mole= 159.54 g/mole ≅ 160 g/mole
- Zn: 65.37 g/mole
- Cu: 63.54 g/mole
- ZnSO₄: 65.37 g/mole + 32 g/mole + 4*16 g/mole= 161.37 g/mole
Then, by stoichiometry of the reaction, the following amounts of mass of reagent and product participate in the reaction:
- CuSO₄: 1 moles* 160 g/mole= 160 g
- Zn: 1 mole* 65.37 g/mole= 65.37 g
- Cu: 1 mole* 63.54 g/mole= 63.54 g
- ZnSO₄: 1 mole* 161.37 g/mole= 161.37 g
Now you can apply the following rule of three: if 160 grams of CuSO₄ react with 65.37 grams of Zn by this reaction stoichiometry, 454 grams of CuSO₄ with how much mass of Zn will it react?
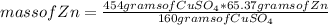
mass of Zn= 185.49 grams
185.49 grams of Zinc would react with 454g (1lb) of copper sulfate