Answer: The quantity of pure
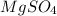 in 2.4 g of
in 2.4 g of
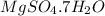 is 1.17 g
is 1.17 g
Step-by-step explanation:
According to avogadro's law, 1 mole of every substance weighs equal to molecular mass and contains avogadro's number
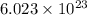 of particles.
of particles.
To calculate the moles, we use the equation:
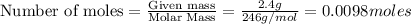
As 1 mole of
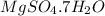 contains = 1 mole of
contains = 1 mole of
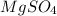
Thus 0.0098 moles of
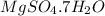 contains =
contains =
 of
of
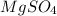
Mass of
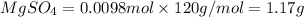
Thus the quantity of pure
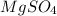 in 2.4 g of
in 2.4 g of
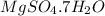 is 1.17 g
is 1.17 g