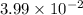The question is incomplete, here is the complete question:
Steam reforming of methane
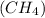 produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a 125 L tank with 20 mol of methane gas and 10 mol of water vapor at 38 degrees celsius. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be 18 mol . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.
produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a 125 L tank with 20 mol of methane gas and 10 mol of water vapor at 38 degrees celsius. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be 18 mol . Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.
Answer: The equilibrium constant for the reaction is
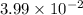
Step-by-step explanation:
We are given:
Initial moles of methane gas = 20 moles
Initial moles of water vapor = 10 moles
Equilibrium moles of carbon monoxide = 18 moles
Volume of the tank = 125 L
The chemical equation for the reaction of methane and water vapor follows:
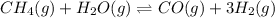
Initial: 20 10
At eqllm: 20-x 10-x x 3x
Evaluating the value of 'x':
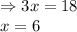
So, equilibrium moles of methane gas = (20 - x) = [20 - 6] = 14 mol
Equilibrium moles of water vapor = (10 - x) = [10 - 6] = 4 mol
Equilibrium moles of carbon monoxide gas = x = 6 mol
The expression of equilibrium constant for the above reaction follows:
![K_(eq)=([H_2]^3[CO])/([CH_4][H_2O])](https://img.qammunity.org/2021/formulas/chemistry/college/hkxyb8fpb9bevygrf85ccs0iqyti9pzxu9.png)
We are given:
![[H_2]=(18)/(125)=0.144M](https://img.qammunity.org/2021/formulas/chemistry/college/qnm1y1nbm23sfw3ypuqjy96o3ozto9cub3.png)
![[CO]=(6)/(125)=0.048M](https://img.qammunity.org/2021/formulas/chemistry/college/49c8g6i9fy0xetjlrux1o6jm7ali4u4oy0.png)
![[CH_4]=(14)/(125)=0.112M](https://img.qammunity.org/2021/formulas/chemistry/college/jihr5t4c9t80qouz8jn6tmtjx158eq9n0l.png)
![[H_2O]=(4)/(125)=0.032M](https://img.qammunity.org/2021/formulas/chemistry/college/z4zvhiwjf5ey7blzwbw6ao21n7l8r4tnyz.png)
Putting values in above expression, we get:
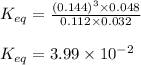
Hence, the equilibrium constant for the reaction is