Answer:
The new wavelength is 112.5 nm.
Step-by-step explanation:
It is given that,
When light with a wavelength of 225 nm is incident on a certain metal surface, electrons are ejected with a maximum kinetic energy of 2.98 × 10⁻¹⁹ J. We need to find the wavelength (in nm) of light that should be used to double the maximum kinetic energy of the electrons ejected from this surface.
The energy of incident electron is given by :
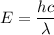
New energy is 2 E and new wavelength is
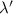 . So,
. So,
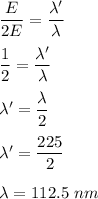
So, the new wavelength is 112.5 nm.