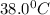Answer: The final temperature is
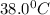
Step-by-step explanation:
The quantity of heat required to raise the temperature of a substance by one degree Celsius is called the specific heat capacity.
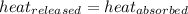
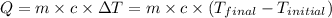
![-[m_1* c_1* (T_(final)-T_1)]=[m_2* c_2* (T_(final)-T_2)]](https://img.qammunity.org/2021/formulas/chemistry/college/r415gvr8ppfjm6b6z5sw2bh44ko5buh8v7.png)
![-[m_1* (T_(final)-T_1)]=[m_2* (T_(final)-T_2)]](https://img.qammunity.org/2021/formulas/chemistry/college/ofo0ox8drb6bamn404suu9cyjczjskkz5a.png) (as
(as
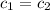 )
)
Q = heat absorbed or released
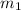 = mass of water at
= mass of water at
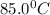 =
=
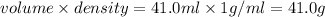
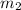 = mass of water at
= mass of water at
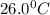 =
=
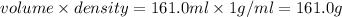
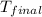 = final temperature = ?
= final temperature = ?
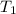 = temperature of 41.0 ml of water =
= temperature of 41.0 ml of water =
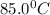
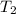 = temperature of 161.0 ml of water =
= temperature of 161.0 ml of water =
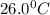
Now put all the given values, we get
![-[41.0* (T_f-85.0)^0C]=161.0* (T_f-26.0)^0C](https://img.qammunity.org/2021/formulas/chemistry/college/jwoqmw4j05788ga8h446poovilx6r2h7bi.png)
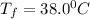
Thus the final temperature is