Answer:
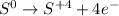
Step-by-step explanation:
Hello,
In this case, for the reaction:
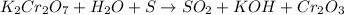
We first must assign the oxidation state of each element:
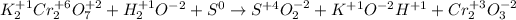
Thus, we should remember that the oxidation half-reaction applies for the element undergoing an increase in its oxidation state, such case is sulfur, for which passes from 0 to +4 as shown below:
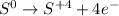
It means, that four electrons were lost due to the effect of the strong oxidizing agent, potassium dichromate.
Best regards.