Answer: The concentration, in moles per litre, of a solution formed when 13.0 g of calcium hydroxide is dissolved in 5.0 L is 0.036 M
Step-by-step explanation:
Molarity of a solution is defined as the number of moles of solute dissolved per liter of the solution.
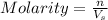
where,
n = moles of solute
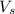 = volume of solution in L
= volume of solution in L
moles of
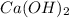 =
=
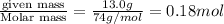
Now put all the given values in the formula of molality, we get
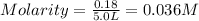
Therefore, the concentration, in moles per litre, of a solution formed when 13.0 g of calcium hydroxide is dissolved in 5.0 L is 0.036 M