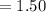Answer:
Number of Nacl units in mass of
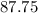 grams
grams
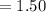
Step-by-step explanation:
Mass of one unit of NaCl or one atom of NaCl is
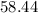 g/mol
g/mol
The total mass of the NaCl is
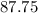 grams.
grams.
The number of unit in
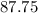 grams of NaCl is equal to
grams of NaCl is equal to
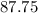 grams divided by mass of one unit of NaCl i.e
grams divided by mass of one unit of NaCl i.e
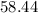 g/mol
g/mol
This can be mathematically be presented as
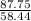 Units
Units
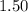 Units
Units
Number of Nacl units in mass of
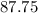 grams
grams