Answer:
B
Step-by-step explanation:
Write the dissociation reaction of solid CoS:
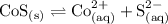
The Ksp expression is hence:
![\displaystyle K_(sp) = [\text{Co}^(2+)][\text{S}^(2-)]](https://img.qammunity.org/2023/formulas/chemistry/college/ttitl2poqci5f496gu425876xndmexeal1.png)
If the molar solubility of CoS is 5.0 × 10⁻²² mol L⁻¹ (M), then 5.0 × 10⁻²² M of Co²⁺ and S²⁻ also forms. Hence, the value of Ksp is:
![\displaystyle \begin{aligned} K_(sp) & = [\text{Co}^(2+)][\text{S}^(2-)] \\ \\ & = (5.0* 10^(-22))^2 \\ \\ & = 2.5* 10^(-43)\end{aligned}](https://img.qammunity.org/2023/formulas/chemistry/college/3nstgie79fhx6f7vrlhyma7cw56od722d4.png)
In conclusion, the answer is B.