Step-by-step explanation:
The electrical force between charges is given by :
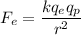
 are charge on electron and proton respectively.
are charge on electron and proton respectively.
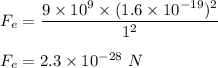
The Gravitational force between masses is given by :
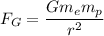
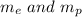 are masses of electron and proton respectively.
are masses of electron and proton respectively.
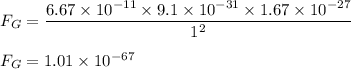
Ratio of electrical to the gravitational force is :
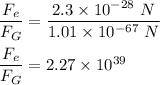
Hence, this is the required solution.