Answer : The molarity of the solution is, 1.1 M LiCl
Explanation : Given,
Mass of
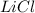 = 32 g
= 32 g
Volume of solution = 655 mL
Molar mass of
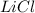 = 42.4 g/mole
= 42.4 g/mole
Molarity : It is defined as the number of moles of solute present in one liter of volume of solution.
Formula used :
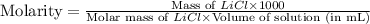
Now put all the given values in this formula, we get:

Therefore, the molarity of the solution is, 1.1 M LiCl