Answer : The correct option is, (4) 6.0 mol
Explanation :
The given balanced chemical equation is,
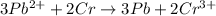
In this reaction, lead undergoes reduction and chromium undergoes oxidation.
Oxidation reaction : It is the reaction in which a substance looses its electrons. In this oxidation state increases.
Reduction reaction : It is the reaction in which a substance gains electrons. In this oxidation state decreases.
Half reactions of oxidation-reduction are :
Oxidation :
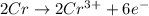
Reduction :
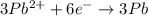
From the reduction reaction, we conclude that 6 moles of electrons gained by the 3 moles of lead ions.
Hence, the correct option is, (4) 6.0 mole