Answer: The mass of
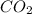 needed is, 0.915 grams.
needed is, 0.915 grams.
Explanation : Given,
Mass of
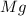 = 1.00 g
= 1.00 g
Molar mass of
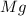 = 24 g/mol
= 24 g/mol
First we have to calculate the moles of
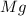 .
.
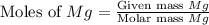
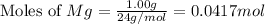
Now we have to calculate the moles of
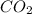
The balanced chemical equation is:
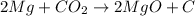
From the balanced reaction we conclude that
As, 2 mole of
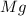 react with 1 mole of
react with 1 mole of
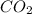
So, 0.0417 moles of
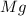 react with
react with
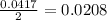 moles of
moles of
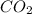
Now we have to calculate the mass of
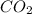
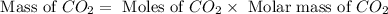
Molar mass of
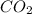 = 44 g/mole
= 44 g/mole
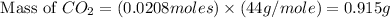
Therefore, the mass of
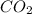 needed is, 0.915 grams.
needed is, 0.915 grams.