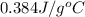Answer : The specific heat of copper is,
Explanation :
Formula used :
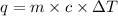
where,
q = heat absorb = 15200 J
m = mass of sample = 105 g
c = specific heat capacity of copper = ?
 = change in temperature =
= change in temperature =
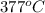
Now put all the given values in the above formula, we get:
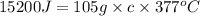
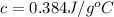
Therefore, the specific heat of copper is,