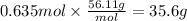Answer:
35.6 g
Step-by-step explanation:
Step 1: Calculate the [H⁺]
We use the following expression
pH = -log [H⁺]
[H⁺] = antilog -pH = antilog -13.90 = 1.259 × 10⁻¹⁴ M
Step 2: Calculate the [OH⁻]
We use the ionic product of water (Kw).
Kw = 10⁻¹⁴ = [H⁺] × [OH⁻]
[OH⁻] = 0.7943 M
Step 3: Calculate the moles of OH⁻
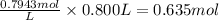
Step 4: Calculate the required moles of KOH
KOH is a strong base that dissociates according to the following equation.
KOH → K⁺ + OH⁻
The molar ratio of KOH to OH⁻ is 1:1. Then, the required moles of KOH are 0.635 moles.
Step 5: Calculate the mass of KOH
The molar mass of KOH is 56.11 g/mol.