Answer: negative , from , to , exothermic
Step-by-step explanation:
An endothermic reaction is a type of chemical reaction in which energy is absorbed from the surrounding. The temperature of the surrounding decreases.
An Exothermic reaction is a type of chemical reaction in which energy is released into the surrounding. The temperature of the surrounding increases.
Endothermic reactions are those in which heat is absorbed by the system from the surroundings and exothermic reactions are those in which heat is released by the system to the surroundings.
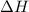 for Endothermic reaction is positive and
for Endothermic reaction is positive and
As on dissolution of salt, the temperature of water increases, the heat has been released from system into surroundings and thus
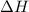 is negative.
is negative.