Answer : The final pressure of gas is, 3.27 bar.
Explanation :
Boyle's Law : It is defined as the pressure of the gas is inversely proportional to the volume of the gas at constant temperature and number of moles.
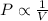
or,
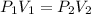
where,
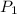 = initial pressure = 4.94 bar
= initial pressure = 4.94 bar
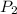 = final pressure = ?
= final pressure = ?
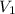 = initial volume = 4.67 L
= initial volume = 4.67 L
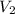 = final volume = 7.05 L
= final volume = 7.05 L
Now put all the given values in the above equation, we get:
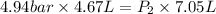
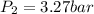
Therefore, the final pressure of gas is, 3.27 bar.